A comprehensive roadmap for MBBS, B.Pharm, M.Pharm, Pharm.D, Life Sciences, Nursing, and AYUSH graduates navigating the global pharmaceutical industry in 2026
USD 158B+
Global drug discovery market by 2034 (CAGR 9.2%)
15,000+
Active job openings in clinical drug development worldwide
₹15–39 LPA
Average salary for drug development professionals in India
10–15 yrs
Average time to bring a single drug to market
1-in-10,000
Molecules that ultimately reach the market from discovery
Every medicine on a pharmacy shelf represents a decade of science, billions in investment and the coordinated work of thousands of professionals, most of whom never wear a white coat in a hospital. A career in drug development sits at the intersection of pharmacology, chemistry, biology, data science, regulatory affairs, and clinical medicine. With the global drug discovery market projected to cross USD 158 billion by 2034 at a 9.2% CAGR, the demand for trained professionals has been increasing.
Yet most medical, pharmacy, and life science graduates in India and across Asia have never been shown a clear map of how to enter this field. University curricula cover pharmacology as academics. They rarely teach how pharmaceutical industries actually develop drugs, what regulators require, or what a clinical research associate does on a Monday morning.
In this blog, we are exactly laying insights on the perception and trying to bring in a positive change. Whether you hold an MBBS, a B.Pharm, a Pharm.D, a life sciences degree, or you are completing an M.Pharm in pharmacology, this actionable roadmap is expertly designed keeping in mind the global standards.
What Is Drug Development and Industrial Pharmacology?
Drug development is the end-to-end process of bringing a new medicine from a laboratory hypothesis to an approved, marketed product. It encompasses drug discovery, preclinical evaluation, clinical trials, regulatory submissions, manufacturing scale-up, and post-market surveillance. A single drug development programme can last 10–15 years and cost over USD 2 billion, including the cost of unapproved trials.
Industrial pharmacology is a subdiscipline that applies pharmacological science within commercial and regulatory settings. In academic pharmacology, you study how drugs act on biological systems. In industrial pharmacology, you use that knowledge to generate data packages that satisfy regulatory agencies, studying how candidate molecules behave in living systems (pharmacokinetics and pharmacodynamics), predicting toxicity before human exposure, designing preclinical safety studies, and writing the pharmacological sections of regulatory submissions.
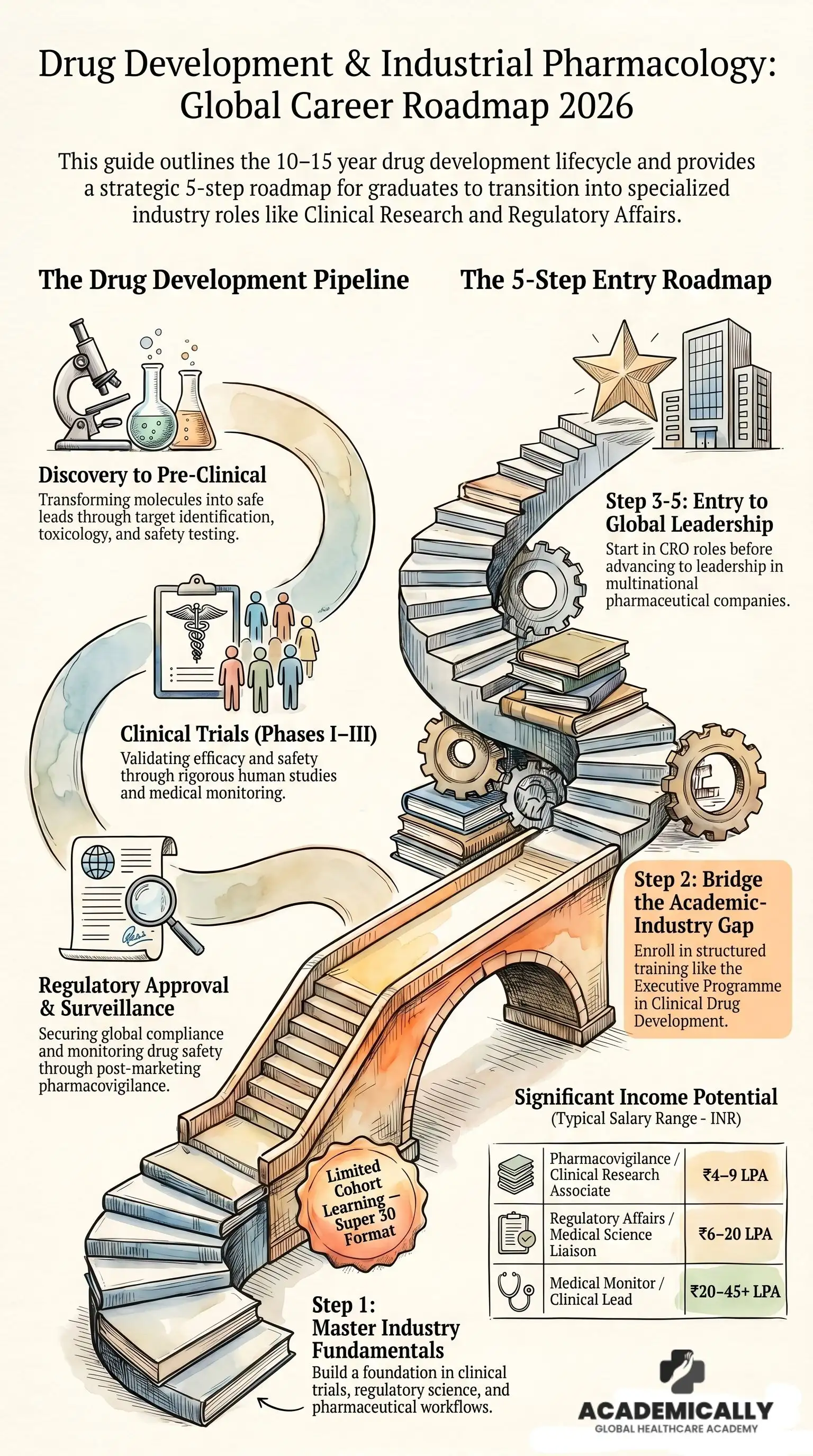
The Drug Development Pipeline: Where Pharmacologists Fit In
The drug development pipeline is the sequence of stages a new compound must pass through before reaching patients. Understanding each stage is not just useful context, it maps directly onto specific roles, skills, and career entry points.
Stage 1
Target Identification & Validation
Biology, genomics, proteomics, AI-assisted
Stage 2
Drug Discovery & Lead Optimisation
Medicinal chemistry, HTS, in silico screening
Stage 3
Preclinical Studies
Safety pharmacology, ADME, toxicology, DMPK
Stage 4
Phase I–III Clinical Trials
CRA, monitors, data management, biostatistics
Stage 5
Regulatory Submission & Approval
Regulatory affairs, medical writing, agency liaison
Stage 6
Post-Market Surveillance
Pharmacovigilance, signal detection, label updates
Pharmacologists are relevant across all six stages. In the preclinical phase, they design and conduct studies to characterise a candidate's safety pharmacology (cardiovascular, CNS, respiratory effects per ICH S7A/S7B guidelines), ADME (absorption, distribution, metabolism, excretion) profiles, and acute/chronic toxicity. In the clinical phases, pharmacologist-trained professionals contribute to clinical trial design, protocol writing, pharmacokinetic sampling strategies, and the integrated summaries of safety in NDA/IND filings.
After approval, pharmacovigilance professionals who have pharmacology or pharmacy backgrounds, monitor adverse event signals in the real-world population. They are far larger and more heterogeneous than any clinical trial.
The most versatile professionals in drug development are those who understand the science behind more than one stage. A preclinical pharmacologist who also understands IND writing is more valuable than one who only knows the bench. A regulatory affairs specialist who has actually run a Phase II trial is a different calibre of candidate from one who has only processed submissions.
Education Roadmap: India and Global Pathways
There is no single degree that unlocks a career in drug development. But there are well-worn educational paths depending on where you are starting. Here is the full picture, including India-specific options that most career guides completely omit.
Undergraduate Foundation
B.Pharm, MBBS, BDS, BSc Life Sciences, BSc Biotechnology, BSc Chemistry, Nursing, any of these provide the scientific foundation. In India, B.Pharm and MBBS are the most common entry credentials.
Focus areas: pharmacology, anatomy, physiology, biochemistry, and practical lab exposure. GPA and laboratory project experience matter more than the specific institution at this stage.
Postgraduate Specialisation
M.Pharm in Pharmacology or Industrial Pharmacology is a direct gateway to preclinical roles in pharma companies and CROs in India. Top institutions: NIPER Hyderabad, NIPER Mohali, NIPER Ahmedabad, Bombay College of Pharmacy, JSS College of Pharmacy (Mysuru and Ooty), BITS Pilani, MAHE Manipal. Pharm.D graduates are well-positioned for clinical pharmacology and pharmacovigilance roles. MD in Pharmacology opens academic, regulatory, and medical monitor pathways for doctors. PhD is valuable for research-heavy discovery roles and academic-industry hybrid positions. MDS and AYUSH postgraduate degrees are relevant for traditional medicine drug development and herbal drug standardisation pathways.
Industry-Specific Executive Training (4–6 Months)
This is the step most Indian graduates skip and then wonder why they are not getting job offers. Academic degrees rarely teach GCP/GLP compliance, IND filing processes, pharmacovigilance database workflows, clinical trial monitoring SOPs, or regulatory submission structure. Structured executive programmes led by industry practitioners bridge this gap more quickly and at a lower cost than a second postgraduate degree. Look for programmes that include capstone projects, real-world case studies, and job-readiness preparation, not just certificates of course completion.

International Credentials (Optional, High-Value)
A Master's in Clinical Research, Pharmacology, or Regulatory Affairs from a US, UK, or Australian university adds global credential weight and often provides internship pipelines into Western CROs or biotech firms.
However, cost-benefit matters, a two-year international Master's costs ₹40–80 lakhs all-in, while a focused 4–6 month executive programme combined with strong internship experience often delivers comparable employability at a fraction of the time and cost for mid-level Indian pharma roles.
The calculus shifts if you are specifically targeting US or EU-based industry entry, where local credentials carry more weight.
Note for AYUSH graduates: BAMS, BHMS, and BUMS doctors are increasingly entering drug development through herbal/botanical drug development, phytopharmacology research, traditional medicine standardisation, and CDSCO's Ayurvedic drug approval pathways. Pharmacovigilance and clinical documentation roles also actively recruit AYUSH professionals. This is an underserved career pathway with growing institutional support from CCIM and AYUSH Ministry initiatives.
Note for Nursing and Allied Health Sciences graduates: Clinical trial coordination, patient recruitment, clinical operations, and some pharmacovigilance roles specifically value nursing and allied health backgrounds. Your clinical understanding and patient-facing communication skills are a genuine competitive advantage that pure pharmacy or life science graduates do not have at entry level.
Step-by-Step Entry Guide to a Successful Clinical Drug Development Career
Build Your Regulatory and Scientific Vocabulary
Before applying for any role, you need to understand how a drug actually progresses through the pipeline and what the regulatory framework around it looks like. This does not require formal enrolment. Read the ICH guidelines: E6(R3) for GCP, S7A for safety pharmacology, M3(R2) for preclinical guidance, E8 for clinical study design.
Study CDSCO's New Drugs and Clinical Trials Rules 2019, especially the Schedule Y requirements. Read FDA and EMA guidance documents for the therapeutic areas that interest you.
This foundation costs nothing and immediately differentiates you in interviews, where the majority of candidates cannot discuss a single regulatory guideline by name or number.
Get Structured Industry Training
Enrol in a programme that teaches the full drug development lifecycle as an integrated system, not just isolated concepts. The best programmes are designed by practitioners who have actually worked in pharma or CROs, include real-world case studies and capstone projects, and cover clinical trials, pharmacovigilance, regulatory affairs, and medical affairs as connected functions.
They should include job readiness preparation. CV writing for pharma roles, interview preparation, and professional communication. This is what hiring managers at Syngene, IQVIA, Dr. Reddy's, and Cipla are looking for and what most formal degrees simply do not teach.
Get Your First Industry Exposure
Log on to Jobslly, India’s first healthcare based job portal. Apply for internships or entry-level positions at pharmaceutical companies, biotech firms, or CROs. In India, CROs are the most accessible entry point. Syngene International, IQVIA India, Lambda Therapeutic Research, Veeda Clinical Research, Synchron Research Services, and Parexel India regularly hire graduates for clinical operations, pharmacovigilance, and regulatory documentation roles.
For research-oriented roles, CDRI (CSIR-Lucknow), NIPER campuses, ICMR-affiliated labs, and the National Centre for Biological Sciences (NCBS) offer postgraduate research exposure. Do not be discouraged by entry salaries at this stage, the experience and network you build in years 1–2 determine your trajectory far more than your starting package.
Build Cross-Functional Experience and Visibility
The professionals who advance fastest in drug development understand adjacent functions. A preclinical pharmacologist who can read a Phase II protocol and identify pharmacological questions is more valuable than one who only knows bench work. Volunteer for cross-team projects.
Ask to sit in on regulatory meetings even if your role does not require it. Attend scientific conferences: ASCPT Annual Meeting, ISPOR India, DIA India Annual Meeting, ClinStar India Conference, Indian Pharmaceutical Congress. Contribute to publications from your research or capstone project, even a conference poster or co-authorship establishes a scientific publication record that opens academic-industry doors later.
Build your LinkedIn presence with analytical commentary on drug approvals, pharmacovigilance signals, or regulatory changes, not just job posts.
Move Into Leadership or Specialisation
Most drug development professionals face a fork. Deepen technical specialisation (becoming a recognised expert in, say, safety pharmacology or pharmacokinetic modelling). You can also move into project/people management (leading clinical trial teams, managing a pharmacovigilance unit, or directing a regulatory affairs department).
Both paths lead to senior compensation, ₹25–45 LPA in India, USD 120,000–200,000 globally, but they require different types of investment. Leadership roles benefit from formal project management training (PMP certification), mentorship programmes, and cross-therapy area exposure.
Deep technical roles increasingly require computational skills and, for pharmacologists, familiarity with population PK/PD software like NONMEM or Phoenix WinNonlin.
Skills That Employers Actually Look For
Bookmark these specific competencies that pharma hiring managers at CROs, Indian generics companies, and MNC pharma consistently request in job descriptions for drug development roles in 2026.
Technical/Scientific (Pre-clinical) Skills
- PK/PD modelling & simulation
- In vitro pharmacology assay design
- In vivo pharmacology (rodent models)
- Safety pharmacology (ICH S7A/B)
- ADME/DMPK studies
- GLP compliance and study management
- Preclinical data interpretation & IND writing
Regulatory & Clinical
- GCP compliance (ICH E6 R3)
- Clinical trial monitoring (SDV, SDR)
- Pharmacovigilance case processing (MedDRA coding)
- Argus Safety/Oracle AERS/VigiBase
- Regulatory writing (IND, NDA, ANDA, CSR)
- CDSCO New Drugs Rules 2019
- EDC systems (Medidata Rave, Veeva Vault)
- Signal detection & benefit-risk assessment
Emerging Skills
- AI-assisted drug discovery (AlphaFold, in silico docking)
- Python/R for pharmacokinetic analysis
- Population PK/PD (NONMEM, Phoenix WinNonlin)
- Digital biomarkers & wearable dataReal-world evidence (RWE) methodology
- Decentralised clinical trial operations
- Pharmacogenomics (PGx) interpretation
- CDISC (SDTM/ADaM) standards
Business, Leadership & Communication
- Scientific and regulatory writing
- Cross-functional team collaboration
- KOL engagement and stakeholder communication
- Presentation of complex data to non-specialists
- Project management fundamentals (PMP)
- Intellectual property and patents basics
- Medical information and literature evaluation
It’s the era to upskill. Learn to work fluently with data. You do not need to become a software engineer, but proficiency in basic pharmacokinetic analysis, and comfort with statistical outputs from clinical trials (p-values, confidence intervals, hazard ratios), puts you in a categorically different tier of candidates.
Pharma is becoming a data-intensive industry at every stage, and professionals who bridge scientific reasoning and data literacy are disproportionately rewarded.
Career Paths, Roles and Salary
Drug development careers are not linear, they are modular. Your background determines which entry point is most accessible; your training and cross-functional experience determine how quickly you move across the grid. Below are the primary career tracks, the key roles within each, and the compensation benchmarks.
Clinical Operations
Clinical Research Associate (CRA)
Monitors clinical trial sites for protocol compliance, data quality, and patient safety. The most accessible entry-level role for B.Pharm, M.Pharm, Life Sciences, and Nursing graduates. High demand across all CROs.
Drug Safety
Drug Safety/Pharmacovigilance Associate
Processes adverse event reports, manages safety databases (Argus, Oracle, Veevo), performs MedDRA coding, and contributes to safety signal detection. One of the largest hiring volumes in Indian pharma and CROs.
Regulatory Affairs
Regulatory Affairs Specialist
Prepares and submits IND, NDA, ANDA, and CTD dossiers; interfaces with CDSCO, USFDA, EMA; manages labelling and compliance. Requires strong regulatory writing and attention to regulatory guideline nuance.
Preclinical Research
Preclinical Pharmacologist/DMPK Scientist
Designs and conducts safety pharmacology and ADME studies; interprets PK/PD data; contributes to IND-enabling pharmacological data packages. It requires M.Pharm in Pharmacology or PhD.
Medical Affairs
Medical Science Liaison (MSL)
Bridges scientific communication between pharma companies and healthcare professionals; manages KOL relationships; generates and disseminates real-world evidence. One of the fastest-growing functions in Indian pharma MNCs.
Clinical Medicine (Industry)
Medical Monitor/Clinical Trial Physician
Physician-led oversight of clinical trial safety; reviews safety reports, investigates SAEs, interfaces with investigators and ethics committees. High compensation. Requires MBBS/MD with GCP training.
Medical Writing
Medical Writer
Produces clinical study reports, regulatory submissions, investigator brochures, and scientific publications. Excellent work-life balance, high precision requirements. Strong demand from both CROs and pharma companies.
Data & Biometrics
Clinical Data Manager/Biostatistician
Manages trial data using EDC systems (Medidata Rave); designs database structures; ensures CDISC compliance. Biostatisticians design statistical analysis plans and interpret trial data. Growing rapidly with decentralised trials.
Salary Benchmark
| Role | Entry Level (₹ LPA) | Mid-Senior (₹ LPA) | Global (USD/yr) |
| Drug Safety / PV Associate | ₹4–7 LPA | ₹10–18 LPA | $55,000–$85,000 |
| Clinical Research Associate (CRA) | ₹5–9 LPA | ₹12–22 LPA | $60,000–$95,000 |
| Regulatory Affairs Specialist | ₹6–12 LPA | ₹15–28 LPA | $75,000–$115,000 |
| Preclinical Pharmacologist | ₹8–15 LPA | ₹18–32 LPA | $80,000–$130,000 |
| Medical Science Liaison (MSL) | ₹12–20 LPA | ₹20–39 LPA | $100,000–$150,000 |
| Medical Writer | ₹6–10 LPA | ₹14–25 LPA | $65,000–$100,000 |
| Medical Monitor / Clinical Trial Physician | ₹20–35 LPA | ₹30–45+ LPA | $130,000–$200,000 |
| Head of Regulatory Affairs / VP | ₹35–60 LPA | ₹50–90+ LPA | $160,000–$250,000+ |
How’s the Indian Market for Drug Development?
India is not a market peripheral to global drug development. It is a structural pillar of it. India accounts for approximately 20% of global generic drug volume by units, produces 60% of the world's vaccines, hosts the second-largest number of USFDA-approved manufacturing facilities outside the US, and runs more clinical trials per year than almost any country outside North America and Western Europe.
The Indian pharmaceutical industry is also home to a rapidly growing CRO ecosystem that has attracted significant global investment over the past decade. Yet most career guidance available to Indian graduates is written for US or UK readers. Here is what you actually need to know to navigate the Indian drug development landscape strategically.
Major Pharma Employers
- Sun Pharma
- Dr. Reddy's
- Cipla
- Lupin
- Biocon
- Zydus Lifesciences
- Aurobindo Pharma
- Torrent Pharma
- Glenmark
- Mankind Pharma
- Divi's Laboratories
Leading CROs in India
- Syngene International
- IQVIA India
- Lambda Therapeutic Research
- Veeda Clinical Research
- Synchron Research
- Parexel India
- ICON Clinical Research
- Medpace India
Key Regulators to Know
- CDSCO (Central Drugs Standard Control Organisation)
- DCGI
- ICMR
- DBT (Dept. of Biotechnology)
- IPA (Indian Pharmacopoeia Commission)
- NPPA
Top Academic/Research Institutions
- NIPER (all 7 campuses)
- CDRI Lucknow
- NII Delhi
- IICT Hyderabad
- JSS Mysuru
- Bombay College of Pharmacy
- BITS Pilani
- MAHE Manipal
- AIIMS New Delhi
Industry Conferences
- Indian Pharmaceutical Congress
- DIA India Annual Meeting
- ClinStar India
- ISPOR India
- PharmaTech Expo
- BioAsia Hyderabad
Pharmacovigilance Infrastructure
- PvPI (Pharmacovigilance Programme of India)
- ADR Monitoring Centres (AMCs) at medical colleges
- National Coordination Centre at IPC Ghaziabad
Why is Industrial Training Important for a Successful Clinical Drug Development Career?
The most consistent feedback from pharma hiring managers in India is not that candidates lack scientific knowledge. It is that they lack the practical, workflow-level understanding of how drug development actually operates in a regulated industry environment. This is the gap that structured executive training is designed to close.
Let’s Hear What the Candidates Have to Say Who Pursued Academically’s Job Assistance Courses
Dr. Saif Haque, MBBS
Resident Medical Officer → Industry Role in Medical Affairs (Bajaj General Insurance, Assistant Manager)
A few months ago, I was actively looking for ways to move into an industry role where I could apply my medical knowledge in a broader, impact-driven environment. That's when I enrolled in the Post Graduate Certificate Programme in Medical Affairs at Academically to upskill. It turned out to be a turning point. What helped the most wasn't just the theory, but the practical exposure, the capstone project, and the feedback sessions with industry experts, including the VP of CIPLA India. I feel fully trained and quite acquainted with what my job is going to be. Thanks to Academically for this course, which is more of an onboarding training than just an academic course.
Dr. Laxmi Bhardwaj, MBBS DCH DNB MBA
Consultant Paediatrician → Pharmacovigilance Course Participant, Academically
From day one, the quality of instruction was outstanding. The faculty brought deep real-world expertise to every session, breaking down complex regulatory frameworks, signal detection methodologies, and case safety reporting with remarkable clarity. You could tell these weren't just educators; they were seasoned professionals who genuinely understood the field from the inside. The learning journey feels smooth and intentional: no confusion, no gaps, just a well-engineered curriculum that builds knowledge progressively. If you're a healthcare professional looking to formalise your pharmacovigilance knowledge, Academically is the place to do it. This is professional education done right.
Ready to Make the Transition?
Academically's Executive Programme in Clinical Drug Development is a 4-month, industry-designed onboarding programme, built for healthcare and life science graduates who want to enter global pharma, biotech, and CRO careers.
To Conclude with…
A career in drug development and industrial pharmacology is one of the most intellectually demanding, globally relevant, and well-compensated career paths available to healthcare and life science graduates today. The work is hard. The timelines are long. The regulatory complexity is real. And the satisfaction of contributing, even incrementally, to the process that delivers a new medicine to patients is unlike most careers.
The field is also more accessible than it appears to most Indian graduates, who have been underserved by career guidance that focuses on the US market, ignores industrial pharmacology as a distinct discipline, and fails to map the specific entry pathways available through India's growing CRO ecosystem and regulatory infrastructure.
The map is now in your hands. The next step is yours.





